We provide expert services for autologous therapies, from collection through manufacturing and back to the patient as quickly and safely as possible.

Autologous Cell Therapy Services
We provide expert services for autologous therapies, from collection through manufacturing and back to the patient as quickly and safely as possible.

Managing collection networks for success
Starting material consistency and quality are crucial for the successful manufacture of autologous cell or gene therapies, making a reliable collection network an essential component of the supply chain.
NMDP BioTherapies assesses centers selected for a partner’s network to ensure they have the infrastructure and capabilities to collect according to our partner’s protocol. We also manage the apheresis centers within our partners’ networks to support ongoing, high-quality autologous collections, while monitoring trends to recommend additional training or process adjustments as needed.
Expertly navigating autologous cell therapy supply chains
Our cell therapy supply chain managers are at the core of our solution. They see the whole picture of every cell therapy order, serving as the single point of contact for a partner and all stakeholders from start to finish. Creating tailored delivery plans, supply chain managers keep all relevant parties up to date on plans and itineraries, including any necessary changes.
NMDP BioTherapies supports the full supply chain for every shipment of an autologous cell therapy, without depending on one single courier. Instead, we maintain relationships with multiple commercial couriers, so that we can choose the best solution for any given logistical challenge.
Extensive experience across the United States and Europe
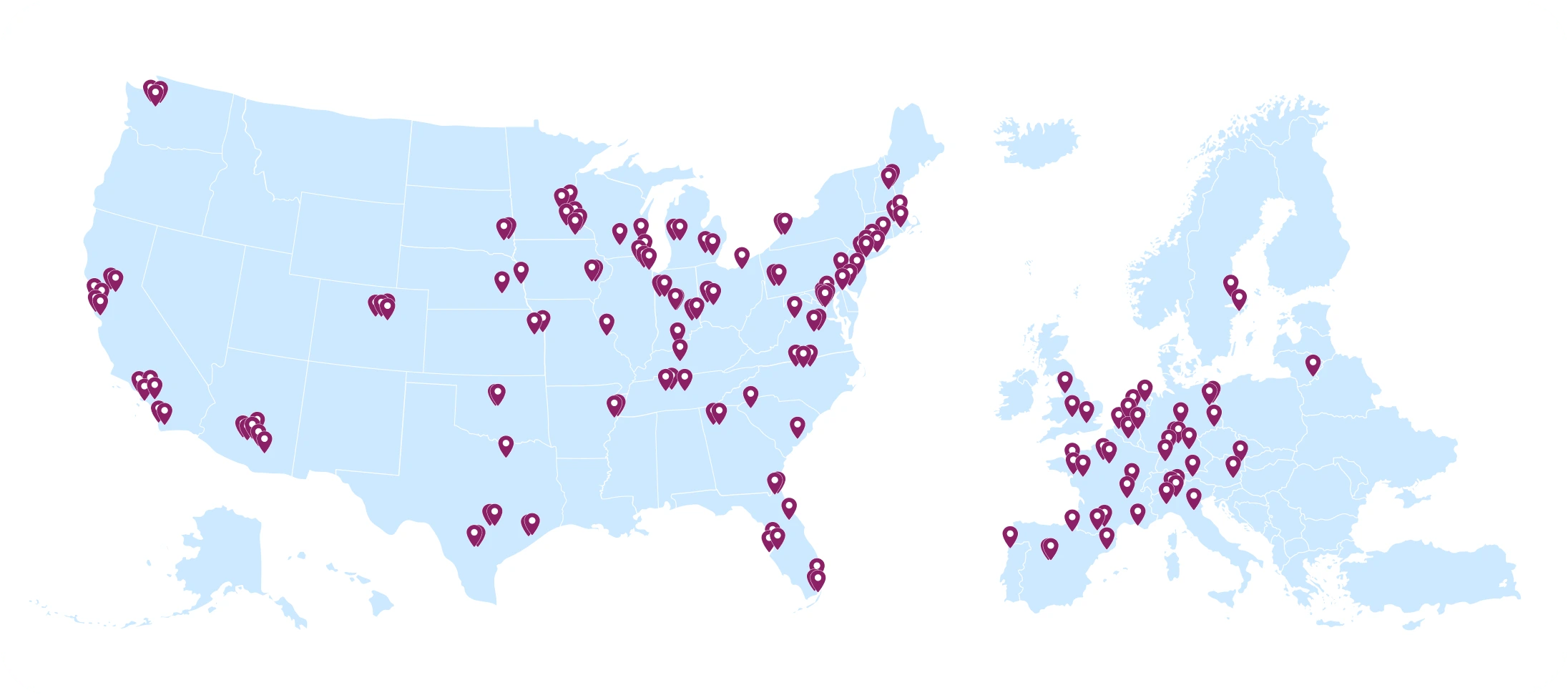
Our NMDP BioTherapies Collection Network team supports our autologous cell and gene therapy clients with onboarding, training and managing apheresis centers across the U.S. and Europe. Each marker on the map represents an apheresis center where our team has performed onboarding, training and/or management.
Extensive experience across the United States and Europe
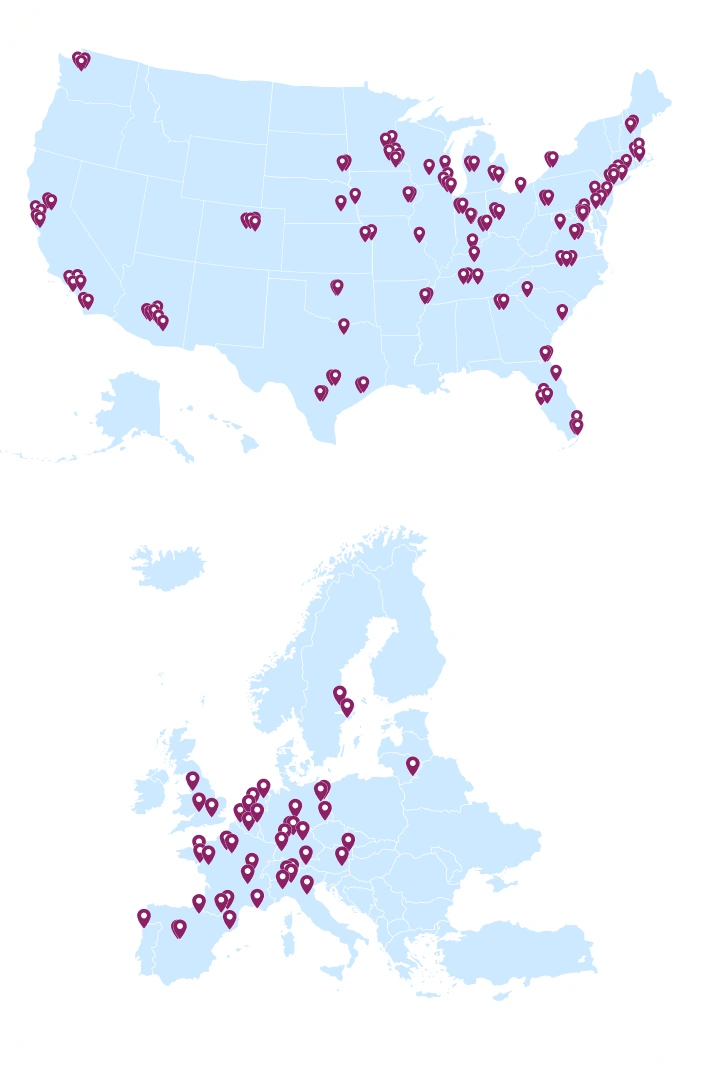
Our NMDP BioTherapies Collection Network team supports our autologous cell and gene therapy clients with onboarding, training and managing apheresis centers across the U.S. and Europe. Each marker on the map represents an apheresis center where our team has performed onboarding, training and/or management.

NMDP [BioTherapies] is truly amazing! As someone new in the world of Cell Therapy, the team has not only helped me understand the logistics and manufacturing process but they have given me reassurance and the confidence of knowing we are receiving the highest quality service and deliverables. They are a huge asset to the team!”

The activities that were outsourced to NMDP [BioTherapies] included site qualification, audits, and all logistics activities. We started off with clinical trials and then also, as we went live and commercial in Europe, we were also able to count on their support as well.”

NMDP [BioTherapies] is the gold standard of logistics in cell-based therapies given the volumes that they manage.”